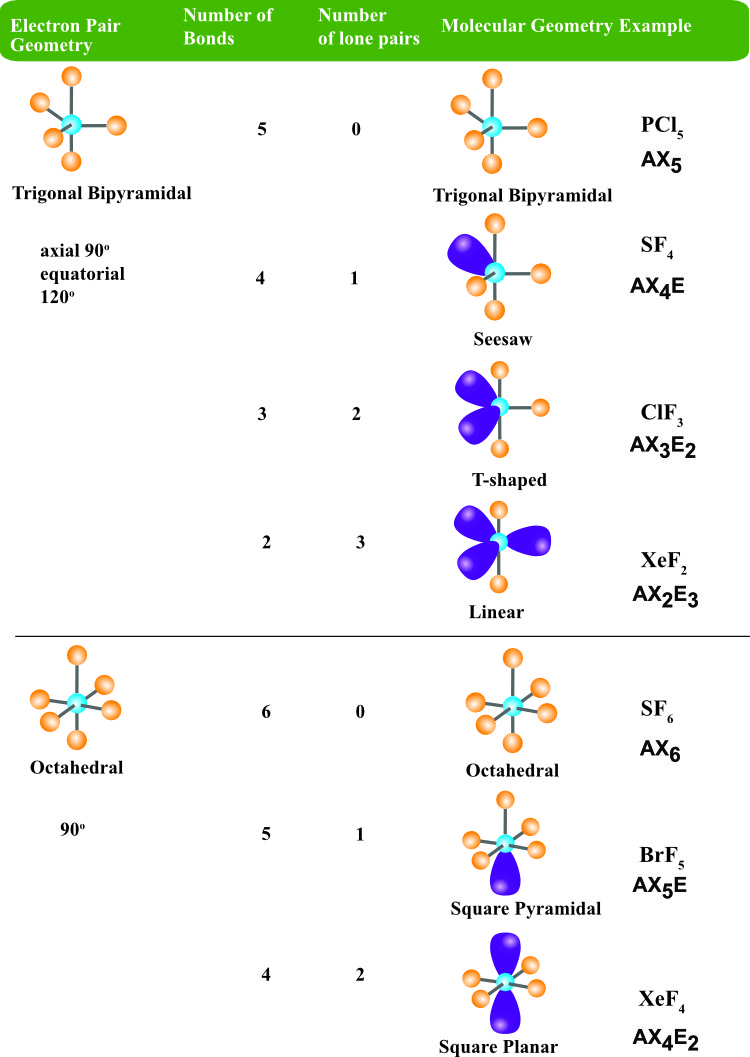
The idealized coordination geometry for eight electron pairs.Ī VSEPR tutorial on the WWW VSEPR tutorial on the WWW, URL: Ĭopyright 1996-2015 Prof Mark Winter. For instance, the idealized geometry for eight electron pairs (epitomized by the anion 2-) is a square antiprism but the energy of other coordination geometries may be very similar or more stable in particular cases. It has tetrahedral electron geometry and a bent molecular shape. The bond angle for H2O2 in its gas phase is 94.8°and has a bond angle of 101.9°. As each Oxygen atom forms an sp3 hybrid orbital, H2O2 has sp3 hybridization. With higher coordination numbers the situation is more complex. There are two lone pairs of electrons on each Oxygen atom thus, there are four lone pairs of electrons for H2O2. The effect of this fluxional process is to average all the fluorine positions. Geometry electron molecular domain angles bond chart domains chemistry lone electrons pairs linear. It is difficult to settle the geometry of the lowest energy configurtion because the geometry of XeF 6 changes rapidly with time, that is, it is fluxional. It gives information about the general shape of the molecule as well as bond lengths, bond angles, torsional angles and any other geometrical parameters that determine the position of each atom. In fact, the structure of XeF 6 is based upon a distorted octahedron, probably towards a monocapped octahedron. Molecular geometry is the three-dimensional arrangement of the atoms that constitute a molecule. These are made up from six bonding pairs and one lone pair. As with IF 7, application of VSEPR rules suggests seven electron pairs. In an H2O molecule, the Oxygen atom forms two single sigma bonds with Hydrogen atoms. The molecule XeF 6 is an interesting case. The molecular geometry of any molecule depends on its Lewis structure, the arrangement of atoms, and its electrons. Iodine heptafluoride, IF 7, is a good example of a pentagonal bipyramidal geometry. The pentagonal bipyramid (left) and the monocapped octahedron. The main geometries without lone pair electrons are: linear, trigonal, tetrahedral, trigonal bipyramidal, and octahedral.  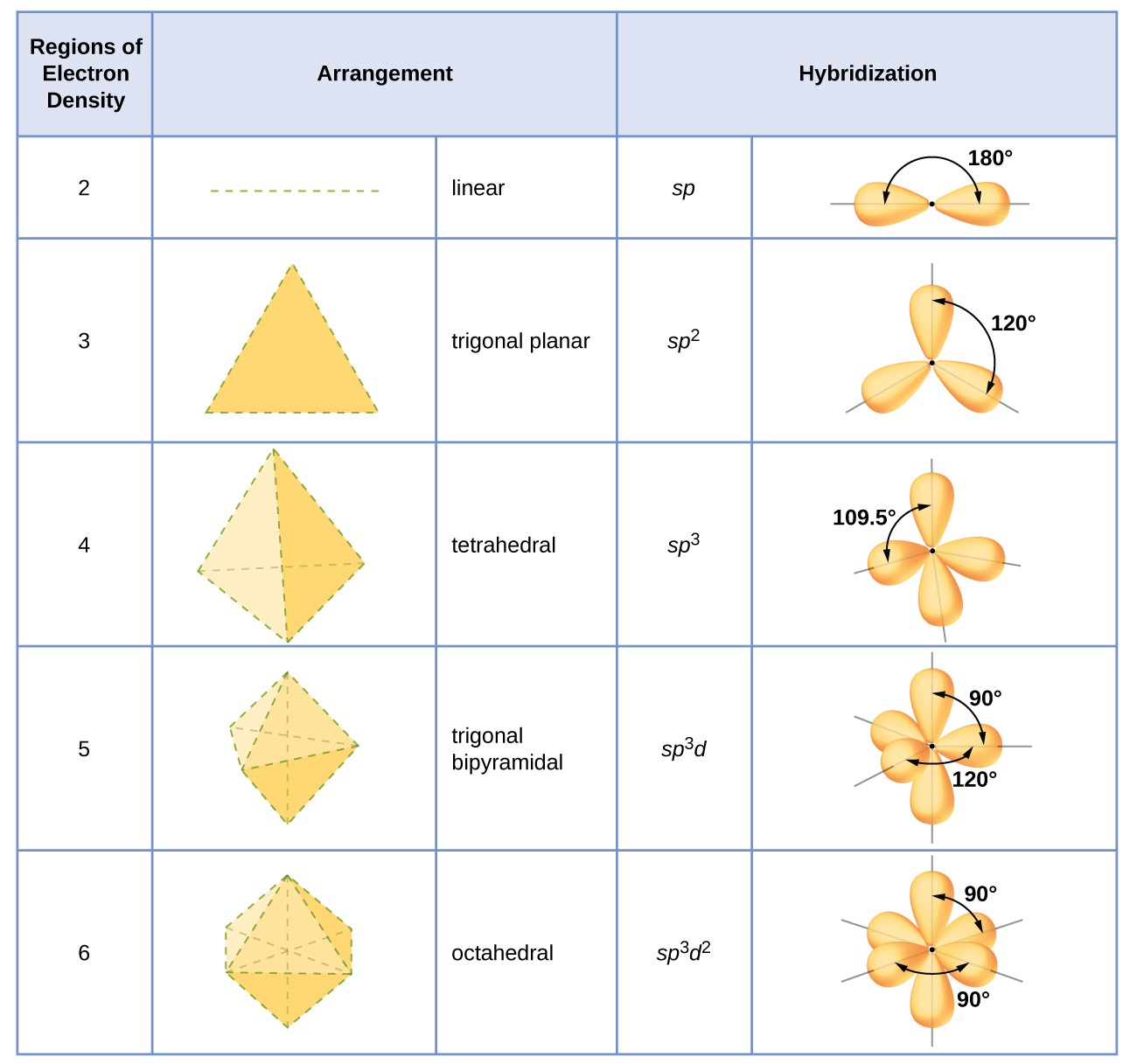
There are several possibilities, including the pentagonal bipyramid and the capped octahedron. It is considerably less easy to draw a distinction between apparently reasonable seven coordinate geometries.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
